Molecular Biomedicine | Targeting regulatory T cells for immunotherapy in melanoma

Open the phone and scan
Regulatory T cells (Tregs) are essential in the maintenance of immunity, and they are also a key to immune suppressive microenvironment in solid tumors. Many studies have revealed the biology of Tregs in various human pathologies. Here authors review recent understandings of the immunophenotypes and suppressive functions of Tregs in melanoma, including Treg recruitment and expansion in a tumor. Tregs are frequently accumulated in melanoma and the ratio of CD8+ T cells versus Tregs in the melanoma is predictive for patient survival. Hence, depletion of Tregs is a promising strategy for the enhancement of anti-melanoma immunity. Many recent studies are aimed to target Tregs in melanoma. Distinguishing Tregs from other immune cells and understanding the function of different subsets of Tregs may contribute to better therapeutic efficacy. Depletion of functional Tregs from the tumor microenvironment has been tested to induce clinically relevant immune responses against melanomas. However, the lack of Treg specific therapeutic antibodies or Treg specific depleting strategies is a big hurdle that is yet to be overcome. Additional studies to fine-tune currently available therapies and more agents that specifically and selectively target tumor infiltrating Tregs in melanoma are urgently needed.

Regulatory T cells (Tregs) are well known to be involved in the immune regulatory activity. Numerous mechanisms of Tregs mediated immune suppression have been investigated in autoimmune diseases, allergy, acute and chronic infection, pregnancy, and cancer. Depletion of Tregs in systemic immunity evokes severe human disease. With the development of technology, immunophenotypes and functions of Tregs in the tumor microenvironment (TME) have been explored. Tregs are identified as an immune suppressor and they have a crucial role in immune suppression in the TME. The relationship of Tregs with other immune cells in the TME provides strategies to target Tregs in the tumor sites. Treg depletion in tumor tissues promotes anti-tumor activity but may also induce fatal immunotherapy-related adverse events. Despite rapid advances in the preclinical and clinical studies, it is still challenging to fine-tune Tregs to promote anti-tumor responses. Here, we review the biological characteristics and immunologic functions of Tregs in the context of immunotherapy. By understanding the role of Tregs in the TME, new therapies targeting Tregs in melanoma may be developed.
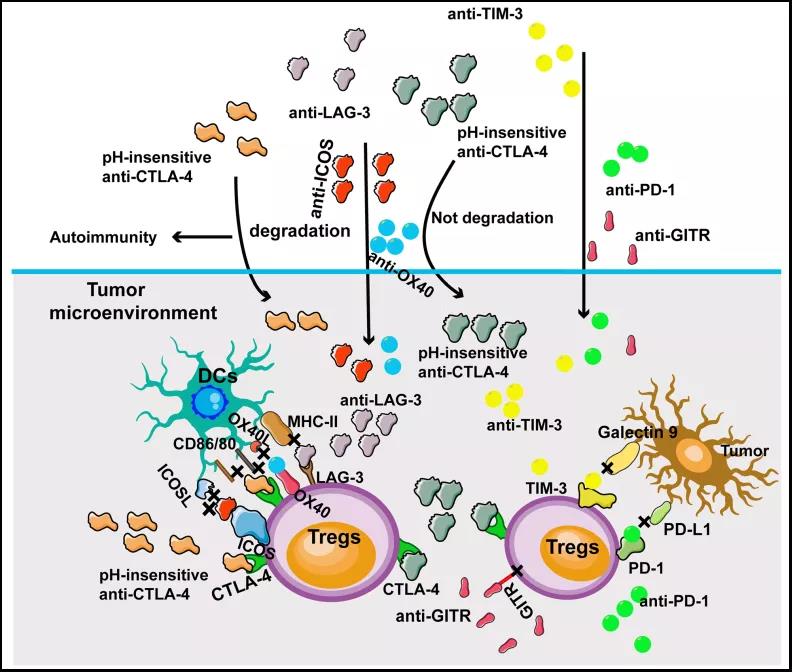
Immune checkpoint therapies targeting Tregs in melanoma
Article Access: https://link.springer.com/article/10.1186/s43556-021-00038-z
Website for Molecular Biomedicine: https://doi.org/10.1186/s43556-021-00027-2
Looking forward to your contributions.


